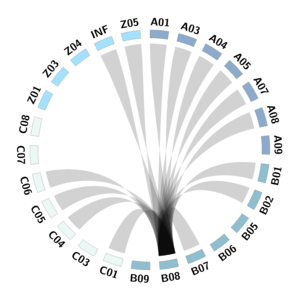
B08
Metabolic characterization of innate lymphoid cells in small cell lung cancer
Despite SCLC high mutational burden, patients poorly respond to T cell-based immunotherapy. Contributing factors are the metabolic barriers in the TME to efficient antitumor response. L. Surace and her team will analyze how innate and adaptive immune cells are influenced by (and influence) the metabolic state of the SCLC tumor microenvironment (TME). Defining the metabolic heterogeneity and the immune composition of SCLC TME, will result in the characterization of innate and adaptive lymphocytes interplay in TME and the identification of new metabolic targets to improve antitumor immunity. We will analyze the phenotype, metabolism, and function of innate and adaptive lymphocytes in treated and untreated SCLC patients with B01, C01, and Z01 and mechanistically dissect their role during tumor progression and metastasis formation using in vivo models and complementing the analysis done by A01 and C05 and A05. Building on the observations made by A05, our team will investigate the metabolic requirements of tumor-infiltrating immune cells and tumor cells in collaboration with B07, in human and murine SCLC, using mass spectrometry. Furthermore, we will use MALDI-imaging to directly measure on tumor sections metabolites produced in the TME and metabolically profile tumor infiltrating lymphocytes. The ultimate goal here is to identify novel routes for therapeutic interven-tion through characterizing the role of immuno-metabolism and the role of innate lymphoid cells.
Principal Investigator

Dr. rer. nat. Laura Surace
Junior Group Leader (STEP program - University of Bonn)
University of Bonn/Uniklinik Bonn
Institute of Clinical Chemistry and Clinical Pharmacology